Electronic Batch Records
Compliance built in, not bolted on.
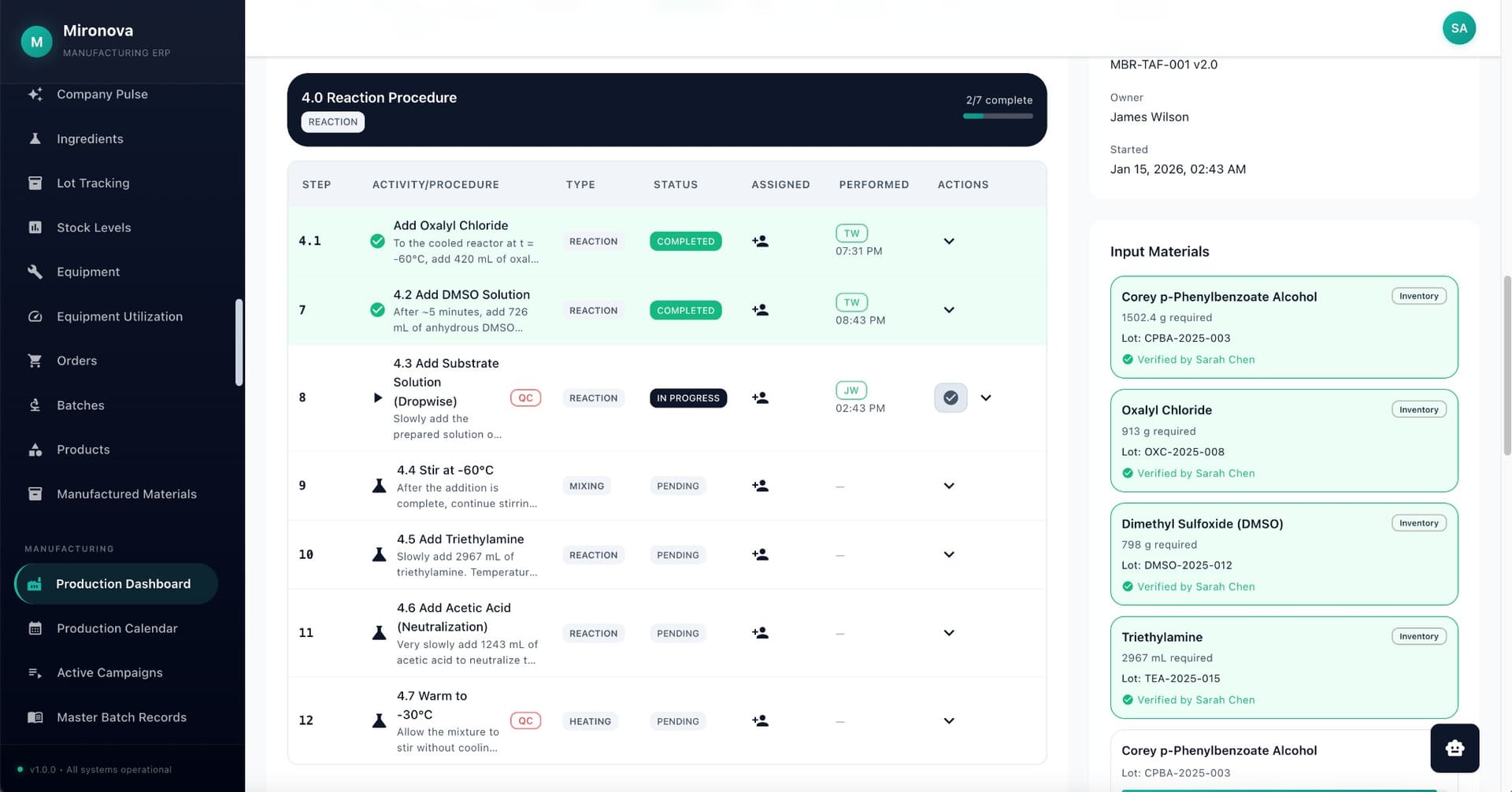
Electronic batch records that operators actually want to use. Step-by-step execution with automatic calculations, in-line deviations, and e-signatures that meet 21 CFR Part 11 requirements. Paper-free manufacturing with complete audit trails.
Capabilities
Everything you need
Step-by-Step Execution
Operators see one step at a time. Can't skip ahead. Can't miss anything.
E-Signatures
PIN verification at critical steps. Meaning captured with every signature.
Automatic Calculations
Formulas calculate automatically. No transcription errors. No math mistakes.
Spec Checking
Values checked against specifications in real-time. Out-of-spec flagged immediately.
In-Line Deviations
When something goes wrong, capture the deviation right there with full context.
Batch Release
One-click release workflow. QA review with full batch history visible.
Zero Data Entry Errors
Operators select from dropdowns, scan barcodes, or confirm calculated values. Equipment data imports automatically. The system prevents errors instead of catching them later.
- Eliminate transcription mistakes
- Reduce batch rejections
- Faster batch release
- Complete audit trail
Zero Data Entry Errors
Built into Helix OS core
Use Cases
Built for real workflows
GMP Manufacturing
Full compliance with pharmaceutical manufacturing requirements.
Multi-Step Synthesis
Complex chemical synthesis with multiple reaction steps and in-process controls.
Formulation & Fill
Compounding, mixing, and filling operations with weight verification.
See Electronic Batch Records in action
30 minutes. No slides. Just the product. See how electronic batch records can transform your operations.