One platform.
Zero silos.
Manufacturing. Quality. R&D. Finance. CRM. Documents.
All connected. All AI-powered. All in Helix OS.
Built by scientists,
for scientists.
We've been making pharmaceuticals for 35 years. We know the pain of juggling 12 different systems that don't talk to each other.
So we built Helix OS to solve our own problems: scattered data, manual paperwork, and the constant question of "where's that file?"
Now we're sharing it with other manufacturers who are tired of the same headaches.
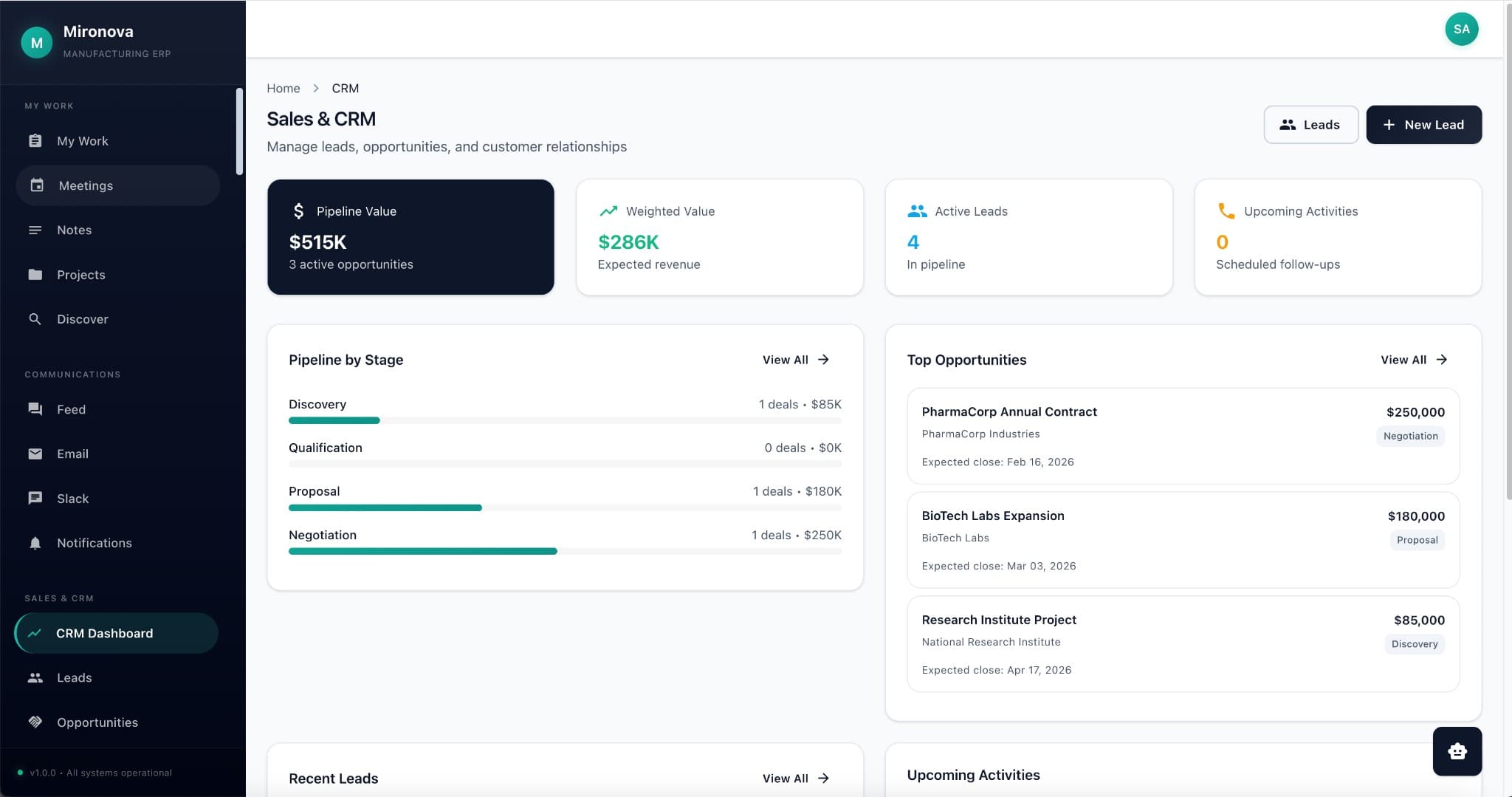
Where software meets science
Piloted & perfected at Mironova Labs
The problem
Sound familiar?
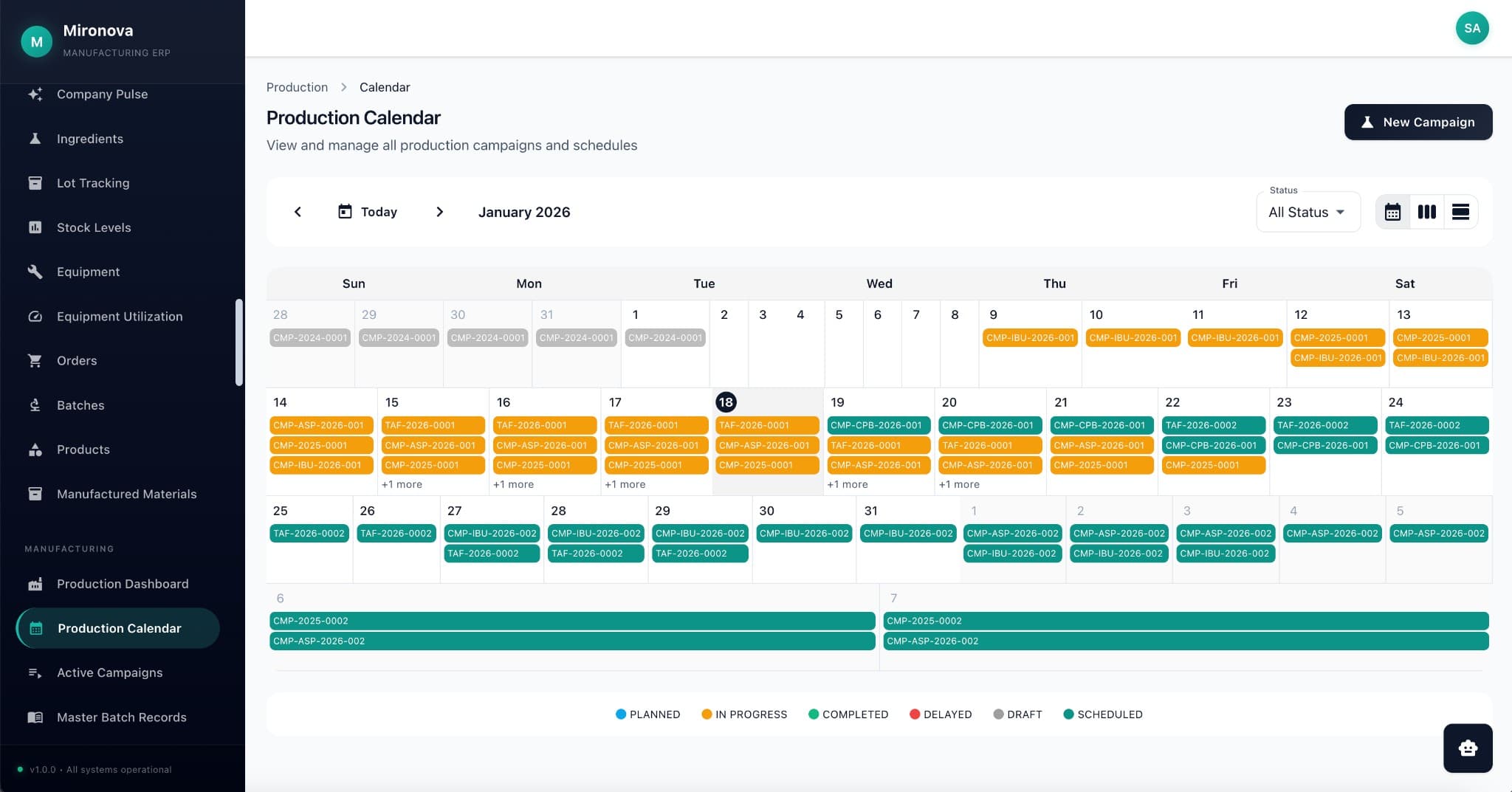
The Platform
Everything connected.
Nothing siloed.
Everything links to everything.
One customer. One product. One batch. One canonical record—with every email, order, file, and note connected. Click any entity, see its entire universe.
Experience the HelixIf only we knew
what we knew.
Your knowledge is scattered. Emails in one place, files in another, batch records in a third. The connections only exist in people's heads—until they leave.
Discovery Feed brings it all together. Pick a customer + a product + a project. See everything related—every email, file, batch, deviation, and order—in one live feed, sorted by what happened most recently.
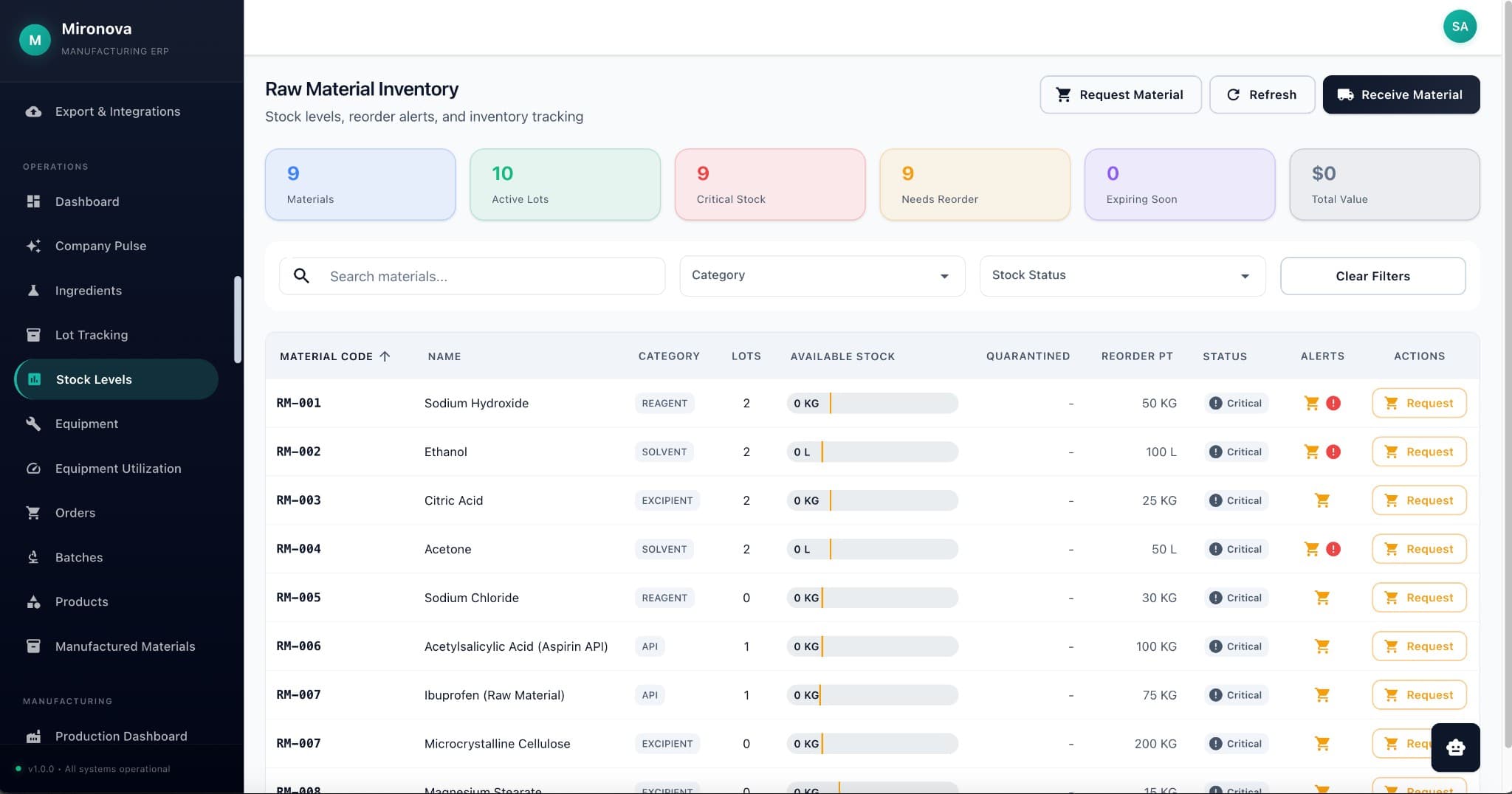
Files aren't trapped in folders. A COA can belong to a customer AND a product AND a batch—all at once. Find it from any angle.
Tag anything to anything. Email someone? It shows in their record. Upload a file? Tag it to customer, product, and project—all at once.
Ask questions with your Discovery feed as context. "Any issues with this customer?" gets a real answer because the AI sees everything.
Add a new customer? The system finds their old emails and files and links them automatically. Your history builds itself.
Enzymatic vs. chemical synthesis comparison study...
Crystalline forms of L-ergothioneine with enhanced stability...
J. Ind. Microbiology, 2024 - Fermentation review...
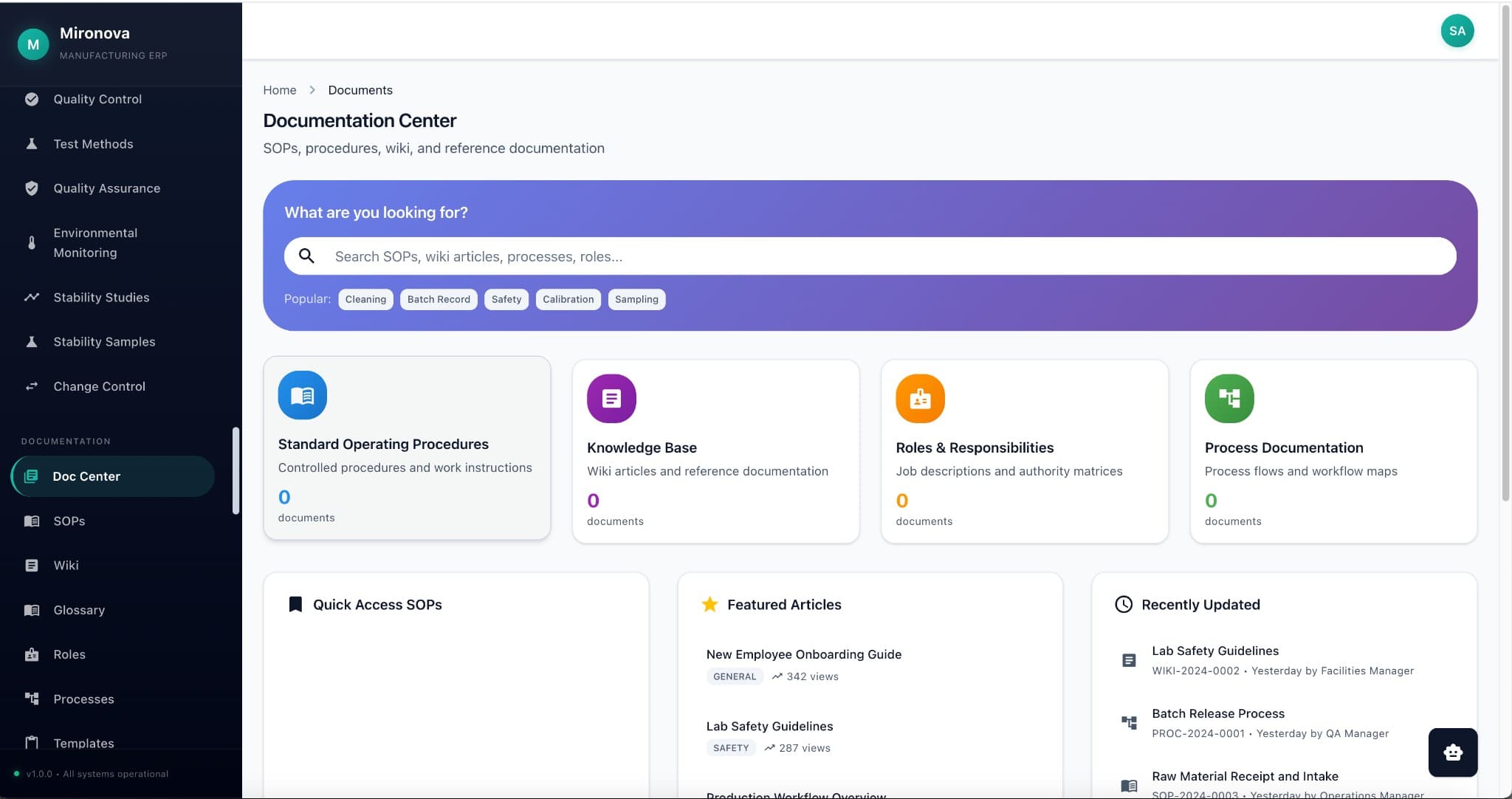
Search everything.
Find anything.
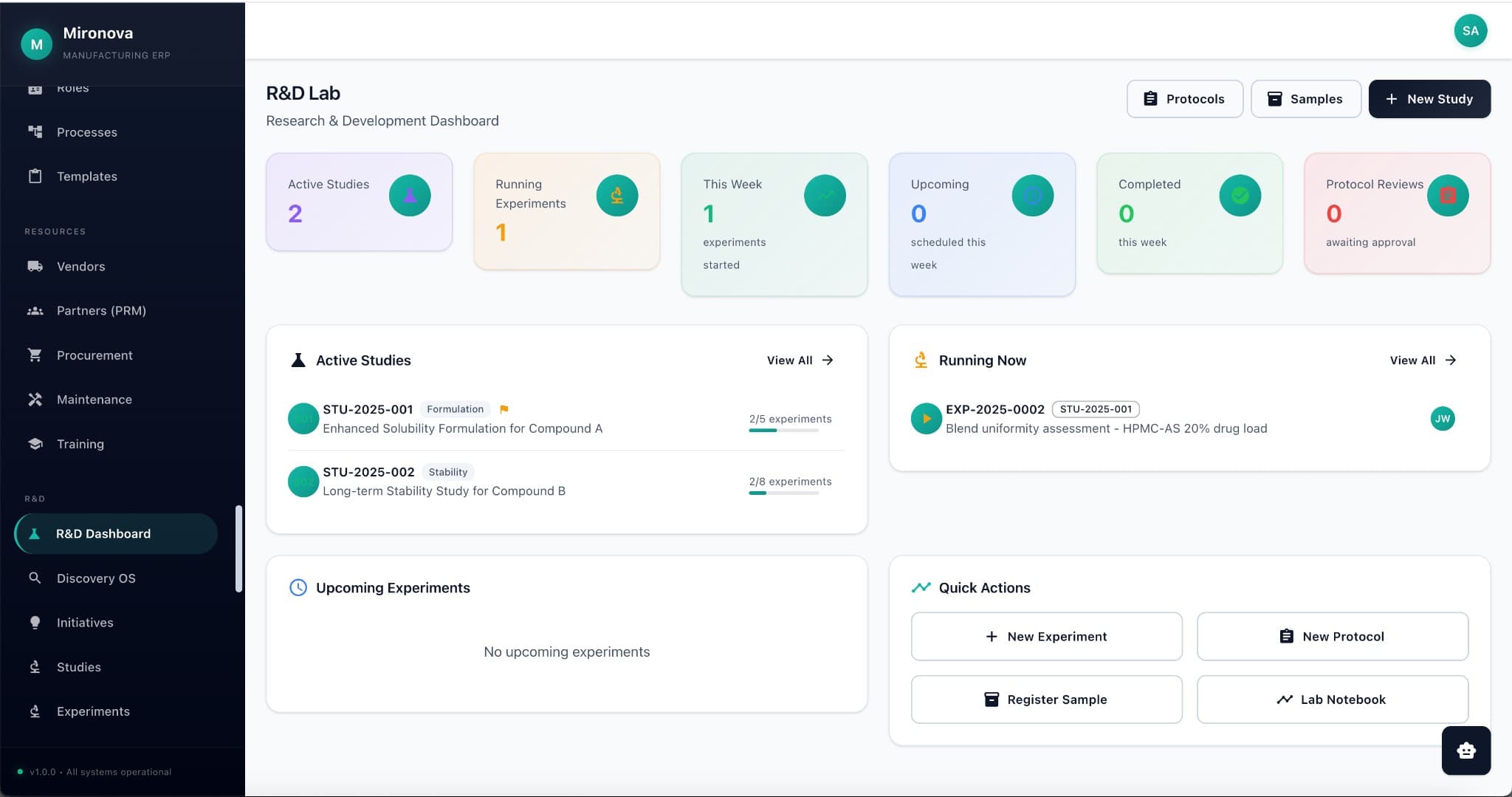
One search across your internal experiments, patents, PubMed, PubChem, and Google Scholar. AI summarizes findings and generates research reports with full citations.
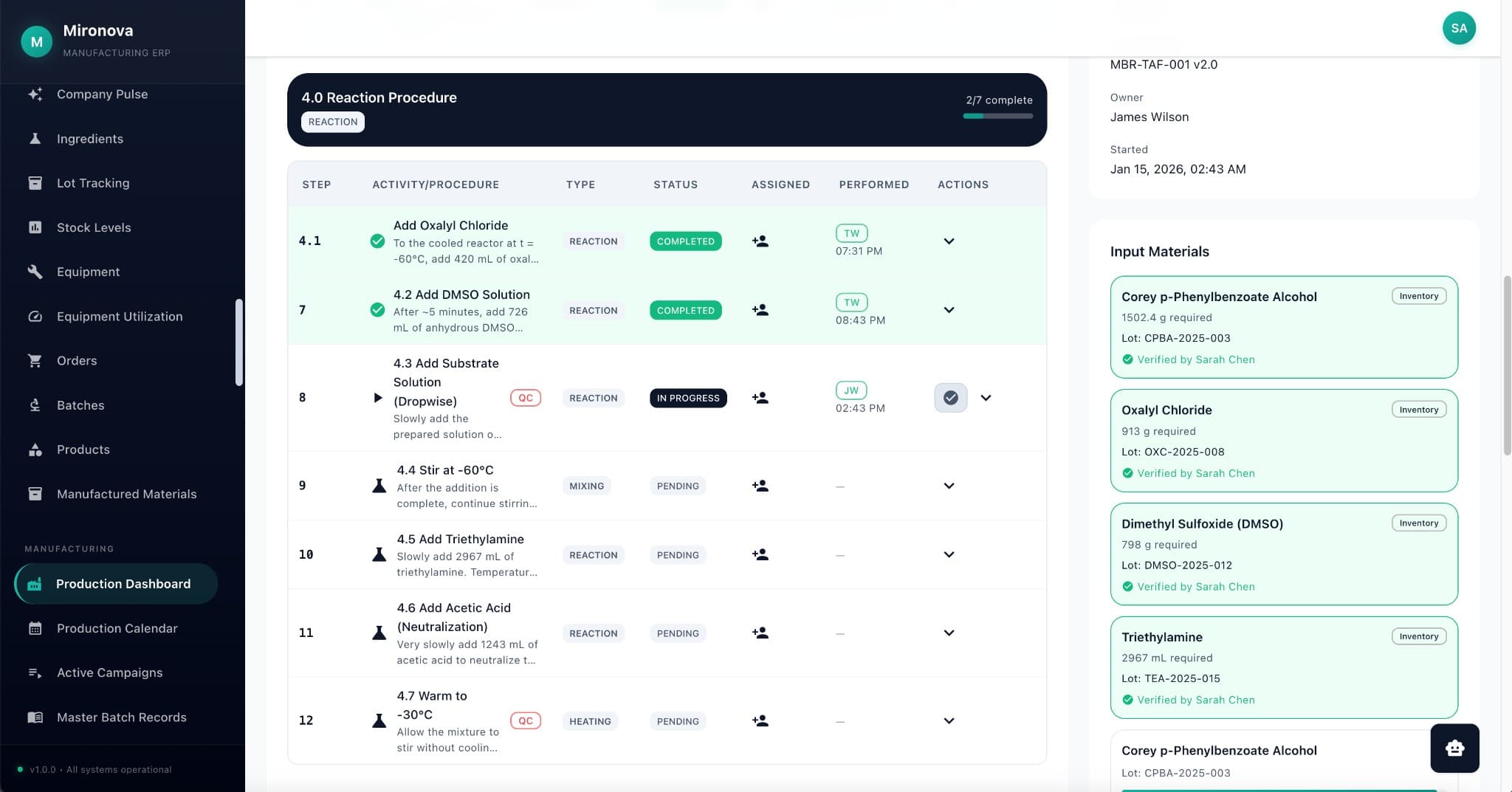
Drop a file. Watch it come alive.
Upload a PDF and see what happens. The AI reads it, classifies it, extracts the data, and links it—all in seconds.
No more "did you refresh?"
Changes show up instantly for everyone. Works offline too—syncs automatically when you're back online.
Ask it anything.
It actually helps.
This isn't a chatbot bolted onto old software. The AI knows your data, understands what you're looking at, and can actually do things for you.
It knows where you are
Ask "any issues?" on a customer page—it knows which customer you mean. No need to explain context.
@ anything
Type @ to pull up any record. Batches, customers, products—just mention them like you would a person.
It does things
Not just answers—actions. Ask it to create a PO, update a record, or generate a report. It actually does it.
No making stuff up
The AI only references real data in your system. It can't hallucinate batch numbers or invent customers that don't exist.
Research mode
Search your internal docs, PubMed, and patents in one query. Get a research report with citations in minutes, not hours.
Respects permissions
The AI can only see and do what you're allowed to. An operator can't ask the AI to approve their own batch.
Built for you
Your role. Your view.
Everyone sees what matters to them. Same data, different lens.
The Difference
We didn't build another ERP.
We built the system we wished existed when we were running production campaigns at 2am.
We run it ourselves
This isn't vaporware. Every feature has been tested in a real manufacturing environment—our own. We eat our own cooking, every day.
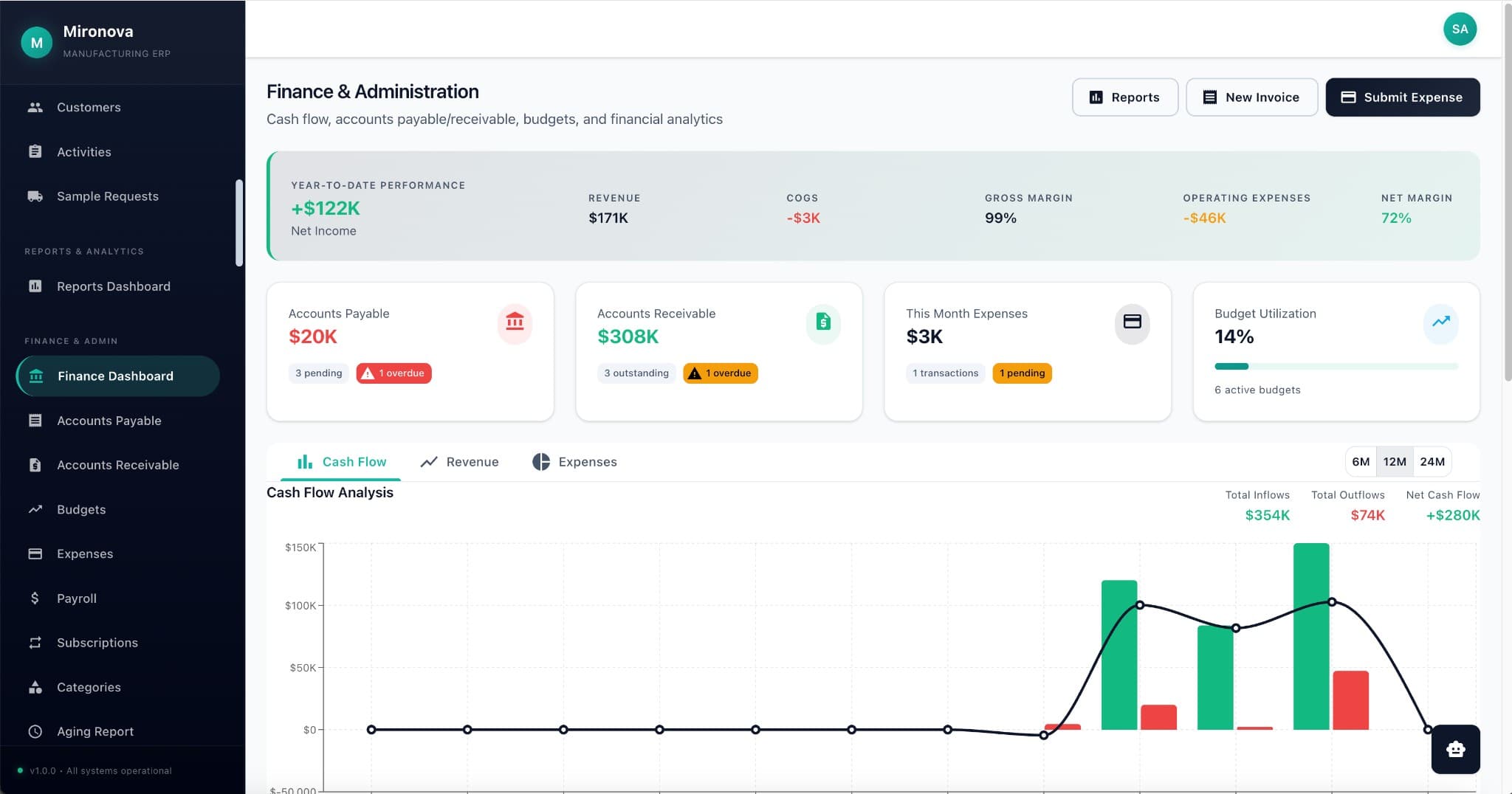
One database. Zero silos.
Production sees what R&D found. Finance sees what shipped. No spreadsheet exports. No "let me check the other system."
AI that actually helps
Not a chatbot. AI that reads your batch records, spots problems early, and drafts your deviation reports.
Compliance built-in
21 CFR Part 11 ready. Full audit trails. E-signatures. Built for regulated industries from day one.
Weeks to go-live, not months
We've done the implementation. We know where it breaks. Our team gets you running fast because we've been in your shoes.
Built for Regulated Industries
Compliance isn't an add-on.
It's the foundation.
21 CFR Part 11 Ready
FDA-compliant electronic signatures with PIN verification, meaning capture, and full audit trails on every action.
- Electronic signatures with declarations
- Dual verification for critical steps
- Complete audit trail retention
- Non-repudiable action logging
Complete Audit Trails
Every action logged with who, what, when, and why. 7-year retention for GxP compliance. Export-ready for audits.
- Immutable change history
- User identity + IP tracking
- Before/after value capture
- One-click audit exports
Enterprise Security
SOC 2 Type II certified infrastructure. Your data encrypted at rest and in transit. Role-based access down to the field level.
- AES-256 encryption
- SSO integration (SAML/OIDC)
- Granular role permissions
- 99.9% uptime SLA
For IT Leaders
Modern stack.
Enterprise grade.
Built on Google Cloud with a modern React + NestJS architecture. API-first design means you can integrate anything. Real-time sync means your team always has the latest data.
Integrations
Plays well with others.
API-first architecture. Connect everything you already use.
Communication & Collaboration
Business & Finance
Storage & Documents
Lab & Scientific Instruments
Developer & AI
Your data. Always yours.
Full data portability. Export everything—records, files, audit trails—in standard formats anytime. Build on top of our API. No artificial barriers.
Don't see your tool? Let's build it together →
What people say
"Finally, it just works."
I picked a customer and a product. Suddenly I could see every email, file, batch, and order between them. All in one view.
Asked about our Taflamide batches. The AI showed me cards for each one that I could click to jump straight there.
Uploaded an invoice from a new vendor. It recognized the vendor, offered to create the record, pre-filled everything.
I was looking at a customer, asked 'any issues?' and it showed me their open deviations. No explaining needed.
Searched for a chemical name. Got hits from inventory, SOPs, emails, and research papers. One search.
We used to spend 3 days preparing for audits. Now it's 10 minutes and a single export.
See it for yourself.
Take the interactive journey, or schedule a personalized demo.
Trusted by pharmaceutical manufacturers, biotech companies, and research institutions worldwide.