CDMT
Also known as: 2-Chloro-4,6-dimethoxy-1,3,5-triazine, 2-Chloro-4,6-dimethoxytriazine, Chlorodimethoxytriazine
High-purity CDMT (2-chloro-4,6-dimethoxy-1,3,5-triazine) — the activated triazine precursor for in-situ DMTMM generation and direct amide bond formation in organic media.
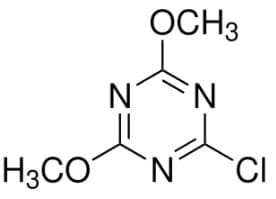
Chemical Structure
Product Information
CAS Number
3140-73-6
Molecular Formula
C₅H₆ClN₃O₂
Molecular Weight
175.57 g/mol
Prices and lead time available upon request
Product Documents
Safety Data Sheet (SDS)
Available on request
Product Specification
Available on request
Certificate of Analysis
Provided with order
Overview
CDMT (2-chloro-4,6-dimethoxy-1,3,5-triazine) is the neutral, activated triazine core that underpins the entire DMTMM product family. It is widely used both as a standalone carboxylic acid activating agent and as the direct precursor for in-situ generation of DMTMM coupling reagents. When combined with a tertiary amine such as N-methylmorpholine (NMM), CDMT rapidly forms the corresponding DMTMM salt in situ, enabling flexible, on-demand access to triazine-based coupling chemistry.
Key Advantages
- Neutral Free Base: No counterion effects — precise stoichiometric control over activation
- Organic Solvent Compatible: Highly soluble in THF, DCM, DMF, and acetonitrile for anhydrous workflows
- In-Situ DMTMM Generation: Combine with NMM or other tertiary amines to generate DMTMM on demand in any desired medium
- Versatile Activation: Activates carboxylic acids directly toward amide bond formation without pre-formed active esters
- Defined Chemistry: Well-characterized, single-component reagent for reproducible results
Applications
In-Situ DMTMM Formation
CDMT is the starting point for generating any DMTMM salt variant in situ. By selecting the tertiary amine partner, chemists can tune the counterion and solubility profile of the resulting coupling species on demand — eliminating the need to stock multiple pre-formed salts.
Peptide Synthesis
CDMT activates amino acid carboxyl groups under mild conditions with minimal epimerization, suitable for both solution-phase and solid-phase peptide workflows.
Polysaccharide & Biopolymer Modification
Effective for coupling small molecules, crosslinkers, or functional groups onto hyaluronic acid, chitosan, alginate, and other biopolymers, particularly in non-aqueous or mixed-solvent systems.
Custom DMTMM Salt Formulations
Used by process chemists who require a specific DMTMM salt variant — including novel counterion combinations — as part of proprietary synthesis routes or regulatory filings.
Technical Information
CDMT is produced through controlled synthesis with rigorous quality control at Mironova Labs. Our material meets pharmaceutical-grade purity standards, ensuring batch-to-batch consistency for demanding applications.
Quality Assurance
- Purity determination by HPLC (≥99.0%)
- Identity confirmation by NMR and MS
- Moisture content analysis
- Batch-to-batch consistency testing
Manufacturing Capabilities
We offer CDMT from research quantities (100 g) to commercial scale (50 kg+) with consistent quality. Custom packaging and labeling available upon request.
Important Safety Information
⚠️ Warning: CDMT is a reactive chlorinated triazine reagent intended for laboratory and manufacturing use only by qualified professionals. Handle with appropriate PPE including gloves, safety glasses, and lab coat. Avoid contact with skin, eyes, and mucous membranes. Keep away from moisture and protic solvents prior to use. Refer to SDS for complete safety information.
Related Resources
Technical data, product specifications, and application guidance.
Questions About This Product?
Our technical team is available to discuss specifications, custom formulations, and bulk pricing.