Bimatoprost Amide
High-purity bimatoprost amide prostaglandin derivative. Research-grade pharmaceutical intermediate manufactured to pharmaceutical standards for laboratory...
Chemical Structure
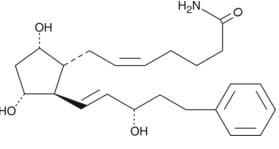
Product Information
CAS Number
155205-89-3
Prices and lead time available upon request
Product Documents
Safety Data Sheet (SDS)
Available on request
Product Specification
Available on request
Product Insert
Available (soon)
Overview
Bimatoprost Amide is a key intermediate and analog of bimatoprost, supplied as a research-grade bioactive compound. This high-purity material is manufactured to pharmaceutical standards for use in pharmaceutical synthesis, laboratory research, and analytical reference purposes.
Product Information
This compound is supplied for cosmetic formulation, research, and manufacturing use by qualified professionals. Intended applications include:
- Pharmaceutical intermediate and synthesis applications
- Laboratory research and analytical reference
- Custom synthesis projects
- Cosmetic raw material supply and formulation development
- In vitro studies and analytical method development
Quality Standards
Manufactured to pharmaceutical-grade standards with complete analytical documentation. Each batch includes Certificate of Analysis with full testing data including:
- HPLC purity determination (≥98%)
- NMR structural confirmation
- Mass spectrometry analysis
- Impurity profiling
Important Safety Information
⚠️ Warning: This is a potent bioactive compound intended for cosmetic, research, and manufacturing use by qualified professionals. NOT for human consumption or self-administration. Regulatory requirements for cosmetic end-use vary by jurisdiction — customers are responsible for compliance in their target markets. Handle with appropriate PPE and safety protocols. Refer to SDS for complete safety information.
Related Resources
Technical data, product specifications, and application guidance.
Questions About This Product?
Our technical team is available to discuss specifications, custom formulations, and regulatory requirements.