Myristoyl Pentapeptide-17
INCI: MYRISTOYL PENTAPEPTIDE-17
Also known as: Myr-Pentapeptide-17, SymPeptide 226EL
A lipopeptide combining a C14 myristoyl fatty acid with a five-amino-acid sequence that upregulates keratin gene expression in hair follicle keratinocytes for lash and brow enhancement.
Chemical Structure
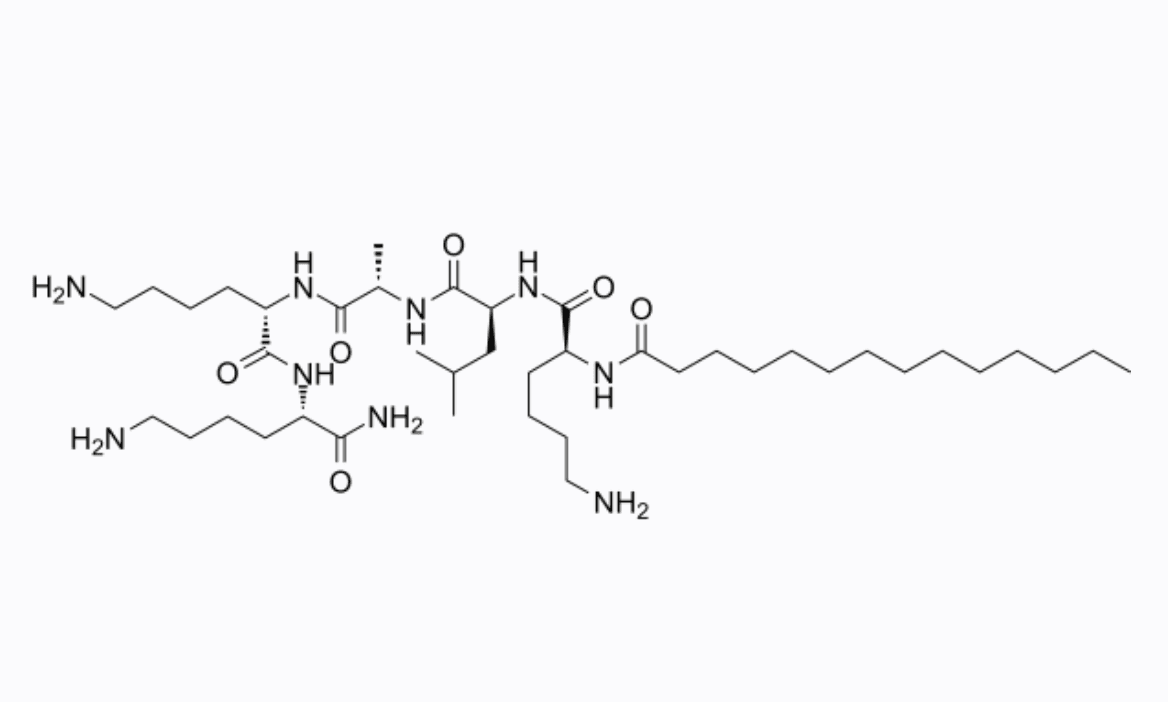
Product Information
INCI Name
MYRISTOYL PENTAPEPTIDE-17
Use Level
1–10 ppm (w/w)
Prices and lead time available upon request
Product Documents
Safety Data Sheet (SDS)
Available on request
Technical Data Sheet (TDS)
Available on request
Certificate of Analysis
Provided with order
Overview
Myristoyl Pentapeptide-17 is a synthetic lipopeptide in which myristic acid (a C14 saturated fatty acid) is covalently conjugated to a pentapeptide sequence. The myristoyl group serves as a penetration enhancer, anchoring the peptide to lipid bilayers and facilitating delivery across the stratum corneum and into the follicular canal. The pentapeptide sequence is designed to signal keratinocytes within the hair follicle to upregulate keratin biosynthesis, supporting the structural integrity and elongation of the hair fiber.
Mironova Labs supplies Myristoyl Pentapeptide-17 as a research-grade cosmetic active, produced in collaboration with a specialized peptide synthesis partner, for integration into scalp serums, lash growth formulas, and eyebrow densification products.
Mechanism of Action
- Keratin gene upregulation — Supplier in vitro data (Symrise, EpDermFT human skin model) indicates the peptide upregulates keratin-associated genes (KRT3, KRT19) and lumican by 80–160%. These results have not been independently replicated in peer-reviewed literature.
- Myristoylation for penetration — The C14 lipid tail increases lipophilicity, enabling the molecule to partition into the follicular lipid environment and improve local bioavailability relative to the free peptide. This is a well-established principle for lipopeptide delivery.
- Elongation phase support — Eyelash clinical studies suggest the peptide supports visible lash elongation. A supplier hair loss study (n=24, 90 days) showed a telogen reduction trend that did not reach statistical significance. Scalp-specific efficacy requires further study.
Formulation Notes
- Active at very low concentrations (1–10 ppm); higher levels do not improve efficacy and are not recommended
- Compatible with aqueous serum, gel, and emulsion bases
- Stable at pH 5.0–7.0; avoid highly alkaline conditions (pH > 8)
- May be combined with Biotinoyl Tripeptide-1 or Myristoyl Hexapeptide-16 for a multi-target hair enhancement approach
- Typical application: leave-on scalp serum, lash serum, brow treatment
Regulatory Information
Myristoyl Pentapeptide-17 is listed in the International Nomenclature of Cosmetic Ingredients (INCI) as MYRISTOYL PENTAPEPTIDE-17. Formulators are responsible for compliance with applicable cosmetic regulations in their markets (EU Cosmetics Regulation, FDA voluntary cosmetic regulations, etc.).
Related Resources
Technical data, product specifications, and application guidance.
Questions About This Ingredient?
Our technical team can discuss specifications, sample requests, and custom synthesis options.